|
Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (CH 3 SO 2 H) Count i Weight. This table has a PDF available for printing. It has the same information as the color periodic table without the color groups for the element groups. This table is for people without color printers or just want to save color ink. In Chapter 1, we described Dalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. First, compute the number of each atom in CH 3 SO 2 H: C: 1, H: 4, S: 1, O: 2. Black and White Periodic Table without Names. (2 atoms)(30.973761 amu/atom) = 6.0474 amu The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. You can also use this molarity calculator to find the mass concentration or molar mass. Molecular weight: 78.133 IUPAC Standard InChI: InChI1S /C2H6OS/c1-4(2)3/h1-2H3 Copy. Design Copyright © 2017 Michael Dayah (). Substitute the known values to calculate the molarity: molarity 5 / (1.2 × 36.46) 0.114 mol/l 0.114 M. As mass/volume molarity × molar mass, then mass / (volume × molar mass) molarity. CO 2 has one carbon atom and two oxygen atoms. Convert the expressions above to obtain a molarity formula.Oxygen (O) has an atomic mass of about 16.00 amu.Printed from Molar Mass, Molecular Weight and Elemental Composition Calculator. Carbon (C) has an atomic mass of about 12.01 amu. Molar mass calculator computes molar mass, molecular weight and elemental composition of any given compound.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound. :max_bytes(150000):strip_icc()/GettyImages-175532236-c614b233b7e84d5487cad8b280f365a4.jpg)
For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in C2H6SO: Molar Mass (g/mol) C (Carbon) 2 × 12.0107 24.0214.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol. Elements: Carbon (C), Hydrogen (H), Oxygen (O), Sulfur (S) Molecular weight: 78.129 g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) 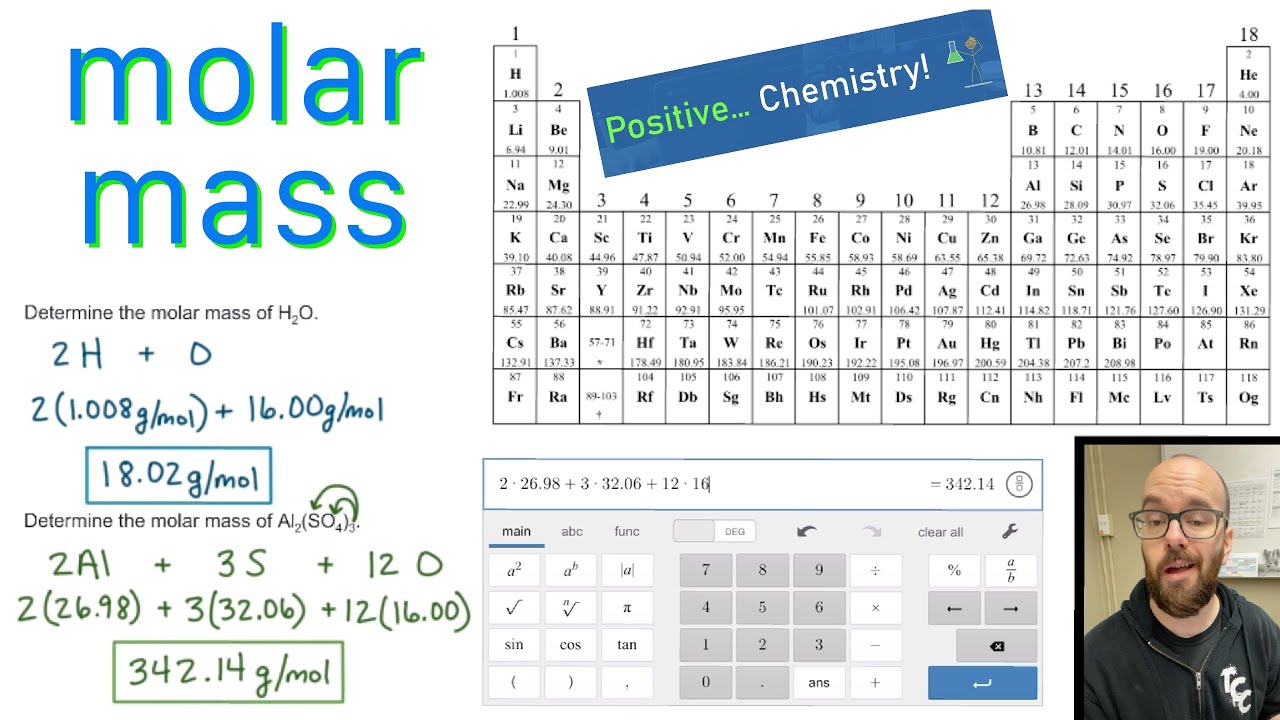
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
